In this interview, News-Medical speaks to Ann-Cathrin Volz and Martin Mangold about Microplate Readers and Recommendations for use.
Can you please introduce yourselves and your role at BMG LABTECH?
Ann-Cathrin Volz:
I am an application specialist for BMG LABTECH, specifically focusing on cell-based assays. After graduating in nutritional sciences at the University of Hohenheim, Germany, I completed my PhD and postdoc in tissue engineering. In my position at BMG LABTECH, I now work on cell culture and related microplate-based assays to support customers in assay optimization for existing and emerging applications.
Martin Mangold:
I also work as an application specialist for BMG LABTECH. My focus is on biochemical assays and interaction studies. During my doctoral studies in pharmaceutical chemistry at the University of Bonn, I screened for specific inhibitors of enzymatic activity. This involved conducting cell culture work and expressing and purifying proteins and antibodies. I now use my knowledge to help BMG LABTECH customers optimize their results.
Can you tell us more about BMG LABTECH and what types of microplate readers the company offers?
Ann-Cathrin Volz:
BMG LABTECH is a microplate reader manufacturer with over 30 years of experience in the field. We are unique because we are solely focused on microplate readers, offering a diverse portfolio to meet various research needs.
Our range includes single-mode to multi-mode instruments, from compact and user-friendly options like the VANTAstar® to high-throughput devices like the PHERAstar® FSX. Each reader is designed to cater to specific research requirements, whether it is absorbance-based ELISAs, very fast and sensitive interaction studies, or solubility measurements using light-scattering.
For instance, our CLARIOstar® Plus is super sensitive and flexible, which is ideal for assay development, while the SPECTROstar® Nano is dedicated to customers with clearly defined absorbance measurement needs.
BMG LABTECH microplate reader portfolio. Image Credit
We also offer accessories like stackers and atmospheric control units for medium to high-throughput applications. With our readers and accessories, we provide a reader control software and a data analysis software called MARS, ensuring a comprehensive solution for researchers.
When measuring with microplate readers, however, there are always factors that need to be taken into account and adjusting screws that can be used for optimisation. These include, for instance, settings on the microplate reader such as the focus height, gain or flash number, but also comprise decisions that have to be made long before the measurement, such as the choice of microplate colour or the selection of the used fluorophore.
How does the choice of microplate colour impact measurements in absorbance, fluorescence and luminescence assays?
Ann-Cathrin Volz:
The choice of microplate colour significantly impacts measurements in absorbance and fluorescence assays. In absorbance measurements, transparent plates are essential to transmit light through the sample. However, the type of transparent plate matters; for example, UV-transparent plates are crucial for applications such as DNA and protein measurements, which are analysed below 300 nm.
Black plates are recommended for fluorescence measurements as they absorb background light, reduce background noise, and improve the signal-to-blank ratio.
White plates, on the other hand, amplify signals. This is advantageous for luminescence measurements which generally show lower emission signal intensities with negligible background signals. White plates provide higher signal-to-blank ratios, enhancing the dynamic window of luminescence assays. Therefore, the choice between black and white plates depends on the assay’s specific requirements and the desired signal amplification or background reduction. The requirements mainly result from the detection mode used.
Can you recommend which plates are best suited for AlphaScreen assays?
Ann-Cathrin Volz:
AlphaScreen is typically classified as a luminescence measurement, thus necessitating the signal amplifying effect of white plates. Nevertheless, cross-talk undoubtedly has an impact as well. Grey plates reduce well-to-well cross-talk and improve assay optimization. Therefore, grey plates are recommended for AlphaScreen.
How do you determine a fluorophore’s optimal excitation and emission wavelengths in a microplate reader?
Martin Mangold:
To determine the optimal excitation and emission wavelengths for a fluorophore in a microplate reader, we examine its excitation and emission spectra. These spectra show where the relative intensities of excitation and emission signals are at their maximum.
To improve the limit of detection, it is crucial to select wavelengths close to these maxima. For example, in the case of the fluorophore in the graph below, we set the excitation and emission wavelengths to 480 and 530 nanometers, respectively.
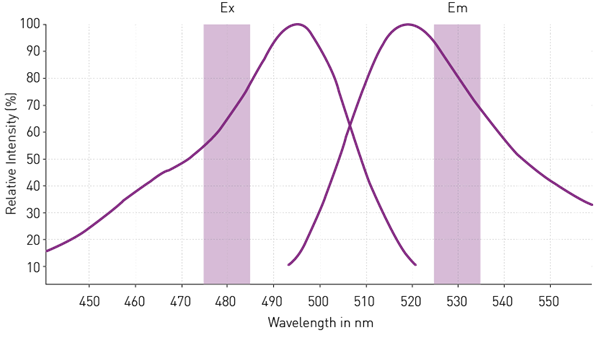
Excitation and emission spectra of a fluorophore. Image Credit
It is also important to have a gap of at least 30 nanometers between excitation and emission wavelengths to prevent the signal transfer from the excitation to the emission channel. This ensures accurate measurements without interference between excitation and emission signals.
How do bandwidth settings affect the sensitivity and accuracy of fluorescence-based assays?
Martin Mangold:
Bandwidth settings play a significant role in the sensitivity and accuracy of fluorescence-based assays. Narrower bandwidths are preferred for excitation wavelengths to specifically excite the fluorophore, as they ensure that only the desired wavelengths are used for excitation, improving specificity and sensitivity.
On the other hand, wider bandwidths may be suitable for collecting more of the emission signal when working with fluorophores or concentrations leading to low signal yields. Here, wider bandwidths can capture more emission signal, potentially increasing sensitivity. However, it is essential to find a balance because excessively wide bandwidths can lead to signal transfer, higher background and loss of sensitivity.
For luminescence-based assays, usually larger bandwidths are beneficial, since more light can be collected from a sample while blank signals remain largely unaffected. Ideally, this results in high signal to blank values.
How does BMG LABTECH’s patented monochromator technology combine flexibility with sensitivity?
Martin Mangold:
BMG LABTECH’s patented monochromator technology offers the flexibility of a monochromator combined with filter-like performance. Traditional monochromators provide flexibility in selecting wavelengths and bandwidths but may sacrifice sensitivity. Filters offer higher sensitivity in comparison to monochromators but are limited to a specific wavelength and bandwidth.
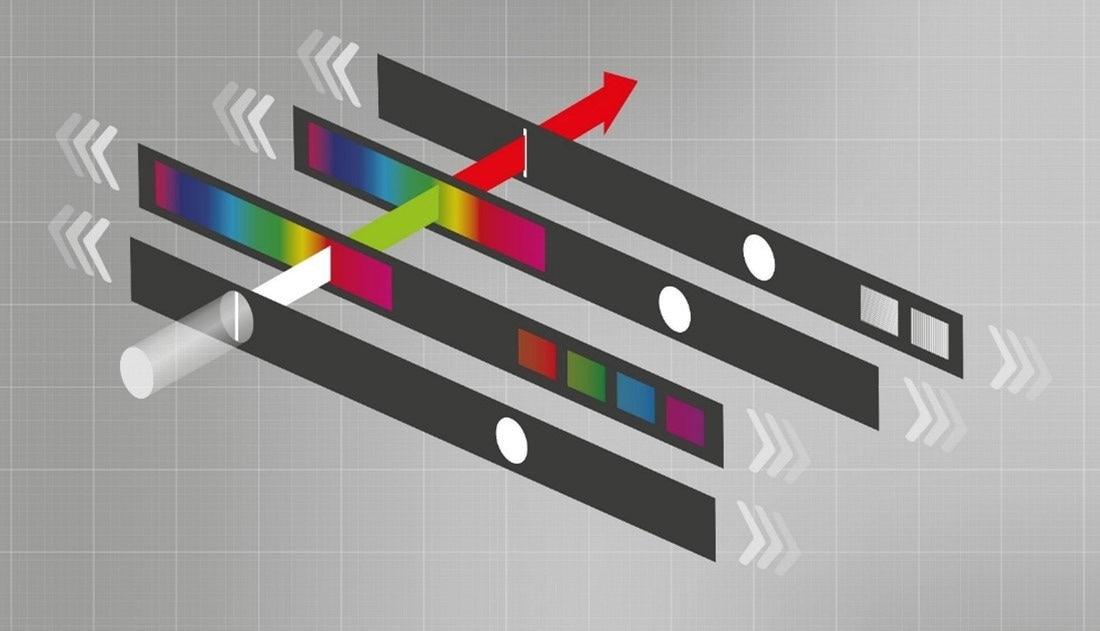
LVF Monochromator schematic: by sliding against each other the LVF slides separate light into distinct wavelengths and bandwidths. Image Credit
Our technology, however, ensures that more light passes through the monochromator, offering enhanced sensitivity. This combination allows researchers to be flexible and sensitive in their measurements, ensuring accurate and reliable results.
Do all filters come with a fixed bandwidth, or is there a certain level of adaptability?
Martin Mangold:
Typically, most filters have a bandwidth of 10 nm but there is a certain degree of customisability. BMG LABTECH offers a wide selection of filter options for specific wavelengths with multiple bandwidth choices. We can provide filters with bandwidths wider than 20 nm for specific wavelengths, ranging up to 100 nm. Filters below 10 nm are also available. The bandwidth of the filter is related to a specific assay and should be discussed with an application specialist.
However, as previously stated, the LVF Monochromators in the VANTAstar and CLARIOstar Plus models offer complete bandwidth flexibility.
What measures can be taken to address autofluorescence in fluorescence-based assays?
Martin Mangold:
Autofluorescence can significantly impact fluorescence-based assays by introducing background noise that interferes with measurements. To address this, adjustments can be made to assay components. For example, switching to alternative assay solutions such as phenol red-free or serum-free cell culture medium, or PBS can reduce autofluorescence and improve the signal-to-blank ratio when running cell-based assays.
Using red-shifted dyes instead of green ones can also minimize autofluorescence, as cellular autofluorescence mainly concentrates in the blue to green wavelength range. These adjustments help enhance the accuracy and reliability of fluorescence-based assays by reducing background noise and improving signal-to-blank ratios.
How do bottom optics help in reducing the negative effects of autofluorescence?
Ann-Cathrin Volz:
With BMG LABTECH readers, measurements can be done either using the top or the bottom optics. The switch from top to bottom optics can actually be done with one click in the software. The types of assays that benefit the most from bottom detection are those where the target is located at the well bottom or where there is unspecific fluorescence in the sample’s supernatant. This is particularly relevant for cell-based assays with adherent cells and cell culture medium containing serum and phenol red. As the light does not have to travel through the supernatant when using bottom optics, signal-to-blank ratios can be increased substantially compared to the use of top optics.
What is focal height, and why is it crucial to adjust the focal height on the brightest sample?
Ann-Cathrin Volz:
Focal height is the distance between the detection system and the microplate or sample and affects the intensity of the measured signal. The optimal focal height is the plane with maximum signal intensity usually found just below the liquid surface. Even slight deviations from the optimal focal height may significantly reduce the signal-to-blank ratio and also the precision of the measurement. Automatic focal adjustment in BMG LABTECH readers works by comparing the signal at different planes and setting the focal height at the peak of the generated curve. This ensures that the ideal focal height is identified and applied for the subsequent measurement. Adjusting the focal height on the brightest sample helps to obtain robust data.
Why is it worth considering scan options and number of flashes for microplate measurements?
Ann-Cathrin Volz:
An optimal flash number as well as the choice of the most suited scan option ensure accurate results to researchers during microplate measurements.
Scan options, impact the thoroughness of data collection. Researchers should select scan options based on the complexity of their assay and desired throughput. Heterogenous cell-based assays, particularly benefit from scan options where multiple measurements are distributed across the well plane in spiral, orbital or matrix patterns and are then averaged subsequently. Besides adherent cell cultures, these options also benefit aggregation assays or microbial growth assays where measurements concentrated at the centre of the well would not be sufficient to represent all cells or the different signal intensities. The use of scan options decreases data variability and at the same time increases reliability and precision.
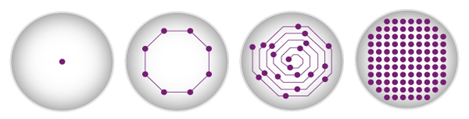
Scan options available with BMG LABTECH microplate readers. Image Credit
Flash number also influences data variability and thereby precision of your measurements and reliability of the generated data. With each flash the sample is illuminated and measured once. When using multiple flashes, an average is formed from the separate measurements. This helps to reduce the effect of outliers which can naturally occur in the measurement process, especially for low-concentration and blank samples. An appropriate number of flashes ensures accurate and reliable data. Typically, 10 to 20 flashes are sufficient for most assays.
It is however essential to balance between accuracy and time expenditure when using scan options and a higher number of flashes, as multiple measurements and plate movements take more time.
Can you explain how adjusting the gain setting influences both signal amplification and measurement range in a microplate reader?
Martin Mangold:
The gain is a factor that defines the amplification of an incoming light signal by the microplate reader detection system and directly influences the intensity of the measured signal. It thereby substantially influences the available measurement range of a microplate reader. A higher gain amplifies the signal to a greater extent, suitable for samples with low signal intensities. Conversely, a lower gain amplifies the signal to a reduced extent and is recommended with high signals.
For instance, in the experiment below, we measured a fluorophore standard curve using different gain values. Choosing the optimal gain setting is important because it ensures that the signal is neither saturated (purple curve) nor too weak to be distinguished from the background (green curve). This underscores the importance of setting the gain appropriately to achieve accurate and reliable results. The optimal gain results in the best signal-to-blank ratio and sensitivity.
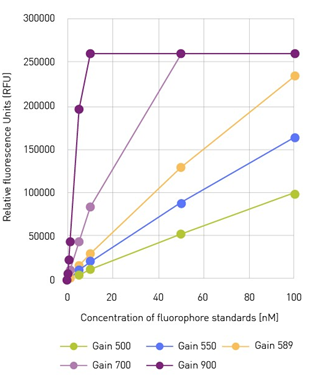
Fluorophore standard curve measured with different gain factors. Image Credit
What is integration time in microplate measurements, and what role does it play?
Martin Mangold:
Integration time in microplate measurements refers to the time window a microplate reader uses to collect light signals from a sample. This parameter is mainly important in luminescence detection, but also in time-resolved fluorescence measurements. The optimal integration time depends on the photon yield of your assay. Longer integration times can be used for assays with a low photon yield over time, while short integration times can be used for assays with high photon yields. Choosing the appropriate integration time is significant because it directly affects the quality and reliability of microplate measurements.
In luminescence-based detection, the appropriate integration time is often determined by the assay type. Flash luminescence shows a very fast decay of emission signal with high signal intensities shortly after the luminescence reaction. Accordingly, a shorter integration time is required. Glow luminescence on the other hand is defined by a slower emission decay but low photon yields, and longer integration times are beneficial.
Our experiments highlighted this by plotting data variability and quality against integration time for glow luminescence samples, showing that longer integration times reduced data variability and increased data quality.
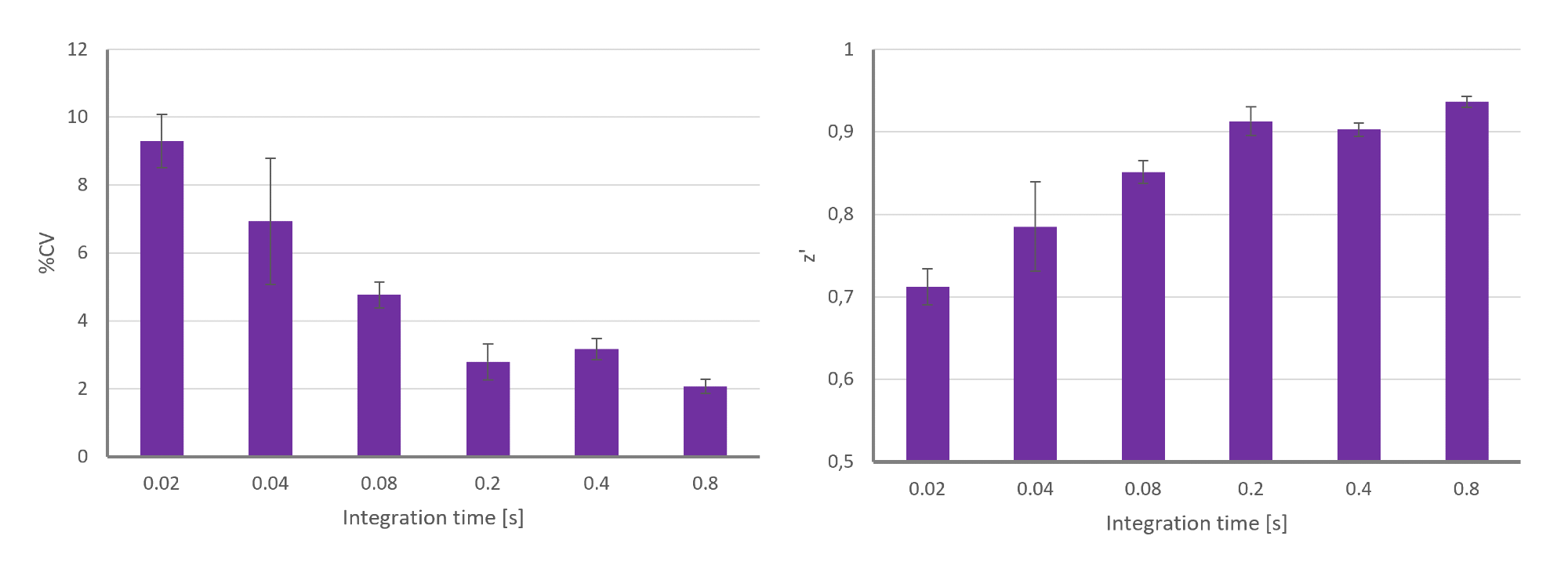
Data variability (%CV= coefficient of variance) and data quality (Z’) plotted for different integration times applied in a glow luminescence-based assay. Image Credit
In the case of time-resolved fluorescence applications, a fluorescent sample will emit light over a longer period of time. Here, the choice of the integration time window should make sure that the sample emits light while the background signal has already subsided.
About Ann-Cathrin Volz
Ann-Cathrin Volz works as Application Specialist for BMG LABTECH with a specific focus on cell-based assays.
After her graduation in Nutritional Sciences at the University of Hohenheim, Germany, she completed her PhD and a Postdoc in the field of Tissue Engineering.
In her position, she can draw on her experience in cell culture and related microplate-based assays to support customers in assay optimisation for existing and emerging applications.
About Martin Mangold
Martin Mangold also works as an Application Specialist for BMG LABTECH. His focus is on biochemical assays and interaction studies.
During his doctoral studies in pharmaceutical chemistry at the University of Bonn, Germany, he screened for specific inhibitors of enzymatic activity, which also included cell culture work, protein and antibody expression, and purification.
He now uses this knowledge to support BMG LABTECH customers in optimising their results.
About BMG LABTECH GmbH
BMG LABTECH has been committed to producing microplate readers for more than twenty years. By focusing on the needs of the scientific community, the company’s innovative microplate readers have earned the company the reputation of being a technology leader in the field.
BMG LABTECH has developed a wide range of dedicated and multi-mode microplate readers for life sciences applications and high-throughput screening.
All BMG LABTECH microplate readers are “Made in Germany” and are conceived, developed, assembled, and tested entirely at our headquarters in Germany.
Since our establishment in Offenburg, Germany in 1989, BMG LABTECH has expanded to offer a worldwide sales and support network with offices in the USA, UK, Australia, Japan and France. Our subsidiaries, regional offices and distributors are committed to bringing you innovative microplate reader technology with the quality and reliability you expect from a German company.
Our staff includes engineers and scientists from the fields of biology, biochemistry, analytical chemistry, and physics.







